Company Outline & Business Model
Company Outline
| Company name | Surv BioPharma Inc. |
| President | Masaki Yamada |
| Locations |
【Head Office & Research Laboratories】 【Tokyo Office】 |
| TEL | 【Head Office】+81(0)99-296-8110 【Tokyo Office】+81(0)3-6824-2310 |
| info(at)survbiopharma.co.jp ※convert (at) into@ | |
| Website URL | https://www.survbiopharma.co.jp |
| Established | August 22, 2022 |
| Capital | JPY1,200,500,000 (including capital reserve) |
| Employees | 11 (including Directors) |
| Core business | Development & marketing of pharmaceutical products |

Our Business Model
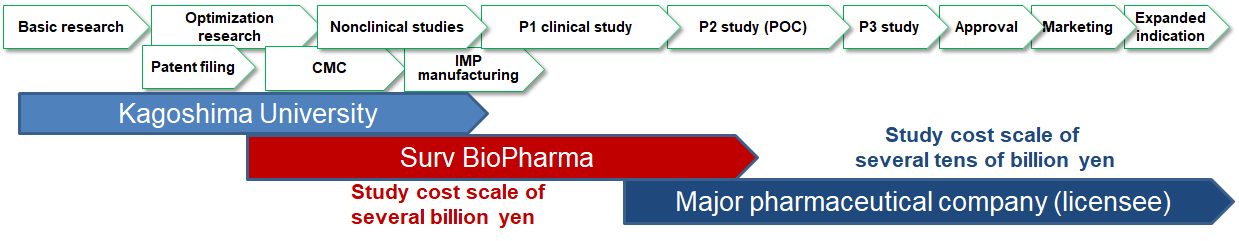
Our pharmaceutical business comprises the in-licensing drug candidates, developed on technologies pioneered by Kagoshima University, under licensing agreements or joint research agreements, and the primarily in-house nonclinical studies and early-stage clinical development of these drug candidates.
After establishing proof-of-concept (POC) (i.e., demonstrating an investigational drug’s efficacy when administered to patients) of our drug candidates as an initial indicator of their commercial value, we will build a revenue model consisting of lump-sum and milestone payments and post-marketing royalty payments through out-licensing to major pharmaceutical companies. These pharmas will then conduct Phase III (P3) studies typically on a cost scale of several tens of billion yen, and will subsequently market the approved drug products.
Through these business activities, Surv BioPharma will create a business structure with sustained and stable profitability, and will contribute to delivering the innovative seed technologies pioneered by Kagoshima University to society.
We will fulfill our corporate social responsibility as a drug discovery venture company originating from Kagoshima University by delivering new therapeutic drugs to patients via major pharmaceutical companies.